We Are The Hair Testing Pioneer & Leader
Psychemedics Corporation is the world’s leading drug testing company using
hair. Psychemedics’ patented hair tests are trusted by Fortune 500 companies, police forces, courts, schools and parents all over the world.
ABOUT PSYCHEMEDICS
Since 1987 we lead the industry for hair testing to detect drugs of abuse. We pioneered the commercial availability of testing for drugs using hair analysis. For that reason, we consistently advance the science. Consequently, our test identifies lifestyle users who exhibit a long-term pattern of drug use.
PATENTED TECHNOLOGY
We pioneered the industry in 1977 after years of research proved that drugs may be detected with hair analysis. Psychemedics developed and patented a process releasing 100% drug held in the hair. As a result, this unique technology detects drug type, and also shows the quantity and historical pattern of use.
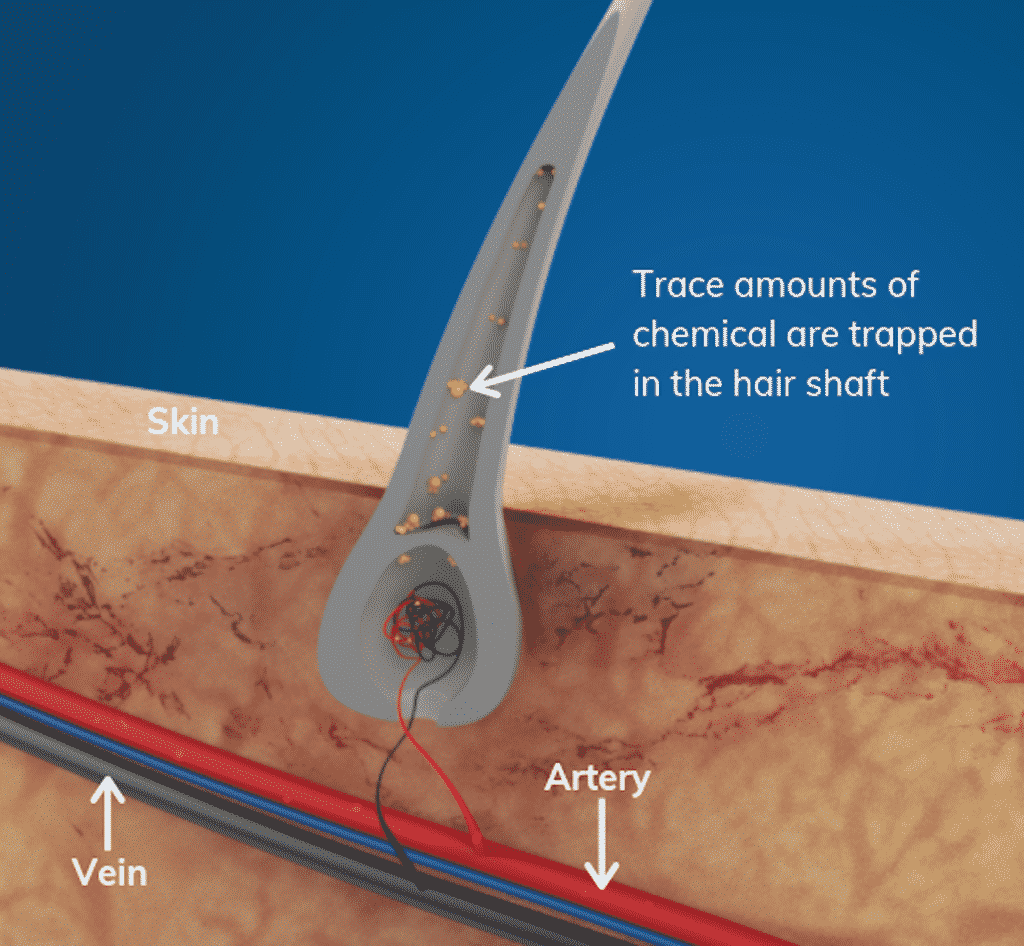
HOW DOES IT WORK?
Ingested drugs circulate in a person’s bloodstream and nourish the developing hair follicle. As a result, the hair shaft traps trace amounts generally proportional to those ingested.
Psychemedics tests for all of the most commonly abused drugs. In addition, we test for the metabolites associated with these drugs, where applicable.
STATE OF THE ART LABORATORY
The FDA awards clearances for all of our screening assays. Equally important, our laboratory holds applicable state licenses and is certified/accredited by several bodies.
- CLIA (Clinical Laboratory Improvement Amendments)
- CAP (College of American Pathologists)
- SO/IEC 17025:2017
ADVANTAGES AND BENEFITS

COLLECTION & TEST
Our standard test requires only a 1.5 inch sample of head hair and provides approximately a 3 month history of drug use. In fact, this is the smallest hair sample collected by any hair testing laboratory.
The collector snips this cosmetically undetectable lock from the back of the head. For that reason collection is sanitary and eliminates embarrassing fluid collections. In addition, Psychemedics offers the only FDA-cleared test for both head and body hair.
Next, the collector seals the sample in tamper evident packaging. Test integrity is critical and for that reason, the donor is present during the entire shipping preparation. Finally, the donor initials the package and it is ready to ship.
After receipt at the laboratory the sample undergoes an initial screen. In the event it screens positive, then it goes through a second test. First, an extensive wash process removes external contaminants. Second, mass spectrometry (GC/MS, GC/MS/MS or LC/MS/MS) confirms the result. And finally, our certifying scientists review the results, which we report to the employer.
